Build a Brain That Squats
Why stress changes your brain chemistry — and what your muscles can do about it
Depression is a complex, multifactorial phenomenon. For decades, it was often framed as a problem of neurotransmitters — especially serotonin, dopamine, and noradrenaline, sometimes with glutamate added to the list.
That monoamine framing shaped modern psychiatry and helped drive the development of widely used antidepressants, including SSRIs. It was a useful model, but not a complete one.
Treatment response varies widely across studies and populations, and many patients do not achieve full remission with first-line medication.
Over time, several consistent observations have challenged the idea that depression can be explained by neurotransmitters alone.
Depressive disorders are not uniform.
Some people develop symptoms early in life, while others experience them later, often alongside metabolic or inflammatory conditions.
Depression frequently coexists with diabetes, metabolic syndrome, and hypertension — conditions that point to broader dysregulation, not just altered synaptic chemistry.
Perhaps more tellingly, certain medical treatments provide a controlled illustration of this link.
Patients receiving interferon therapy, particularly in the context of hepatitis C, can develop depressive symptoms despite having no prior psychiatric history — a phenomenon linked to immune activation and changes in tryptophan metabolism.
In other words, activating the immune system can be enough to alter mood, even when neurotransmitters were not the original problem.
From Neurotransmitters to Metabolism
This shift in perspective has led to a broader understanding of depression — not as a simple chemical imbalance, but as a state emerging from interactions between the nervous system, the immune system, and metabolism.
One pathway sits at the intersection of all three: the kynurenine pathway.
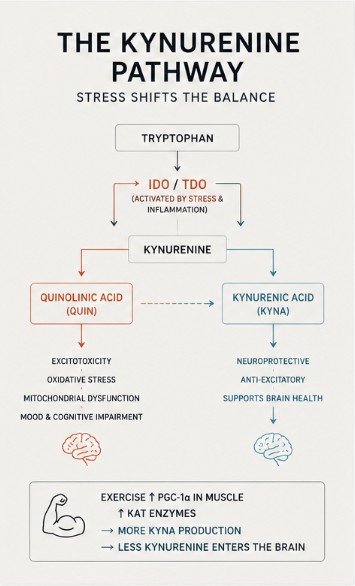
Tryptophan, an amino acid obtained through the diet, is often associated with serotonin production. In reality, most of it is metabolized through the kynurenine pathway, which ultimately contributes to the production of NAD+, a central cofactor in cellular energy metabolism.
Under normal conditions, this routing is expected and necessary. The system is not broken — it is simply wired this way.
The situation changes, however, under stress and inflammation.
When Stress Rewrites the Pathway
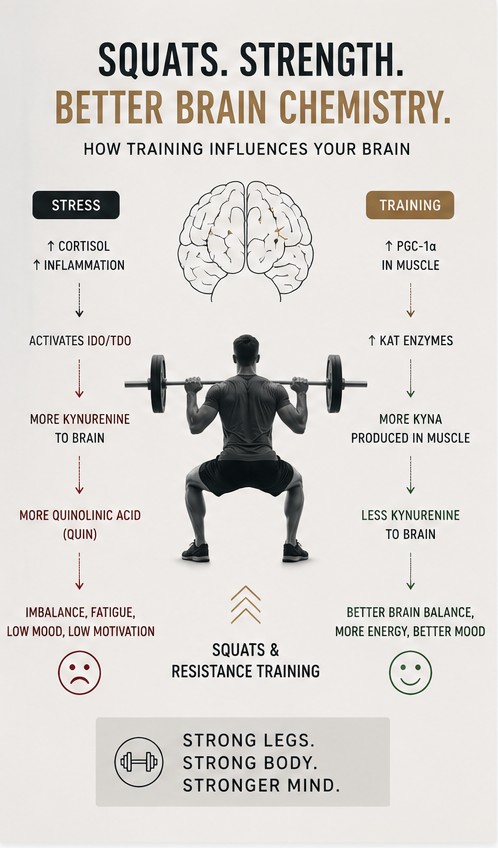
In physiological terms, stress does not simply drain the system — it reorganizes it.
Two enzymes, indoleamine 2,3-dioxygenase (IDO) and tryptophan 2,3-dioxygenase (TDO), act as gatekeepers of the kynurenine pathway. IDO is primarily activated by inflammatory cytokines, while TDO responds to glucocorticoids such as cortisol.
When these signals rise, more tryptophan is diverted into kynurenine metabolism, increasing downstream metabolite production.
You can think of this as a metabolic detour. Under baseline conditions, traffic flows in a relatively balanced way. Under stress, the system starts redirecting cars down a different road, one that leads to a very different neighborhood.
At this point, the pathway begins to matter less as an energy route, and more as a regulator of the brain’s chemical environment.
QUIN: When Excitation Loses Its Brakes
Among these metabolites, quinolinic acid (QUIN) is one of the most biologically active.
QUIN is a neuroactive kynurenine-pathway metabolite with excitatory, NMDA-related effects. In excess, it can contribute to glutamatergic imbalance, oxidative stress, and mitochondrial dysfunction.
 It is a bit like pressing the accelerator while quietly cutting the brake lines.
It is a bit like pressing the accelerator while quietly cutting the brake lines.
The downstream consequences are well characterized: elevated excitatory tone can burden neurons, disrupt energy balance, and contribute to fatigue, reduced motivation, and cognitive difficulty.
Certain brain regions appear particularly sensitive to these effects, notably the hippocampus and the striatum — areas involved in memory, mood regulation, and motivation.
This is not a system failure. It is an adaptive response. But like many adaptive responses, it becomes maladaptive when sustained.
Sickness Behavior
From an evolutionary perspective, many features of depression resemble what is known as sickness behavior.
When an organism is injured or infected, it reduces activity, withdraws socially, and conserves energy.
This coordinated response supports recovery and reduces the risk of further harm or transmission. In a way, the system is temporarily switching priorities — less “perform,” more “repair”.
Similar responses have been observed in humans following immune activation, including after endotoxin exposure or vaccination. Lower mood, reduced motivation, altered sleep, and decreased social engagement are part of this biologically conserved program.
The issue is not the response itself, but its persistence. When inflammatory or stress signals remain elevated, the system continues to operate in a mode that was never meant to be sustained.
Muscle as a Buffer
This is where training becomes particularly relevant.
Skeletal muscle is not just a contractile tissue — it is metabolically active and capable of influencing systemic physiology. Under regular exercise, especially endurance-type training, muscles adapt in ways that directly affect kynurenine metabolism.
Exercise increases the expression of PGC-1α, a regulator of mitochondrial biogenesis. In skeletal muscle, this upregulates kynurenine aminotransferases (KATs), enzymes that convert kynurenine into kynurenic acid (KYNA).
Unlike kynurenine, KYNA does not readily cross the blood-brain barrier.
This creates a peripheral sink, where kynurenine is metabolized in muscle before it can enter the brain and contribute to the production of more reactive metabolites. In practical terms, your muscles help shape what reaches the brain in the first place.
Building the System
Different forms of training contribute to this process in complementary ways.
Aerobic exercise supports mitochondrial density and enhances the enzymatic machinery involved in kynurenine metabolism. High-intensity intervals provide a more condensed stimulus, while resistance training supports overall metabolic health and preserves the structural capacity of muscle.
Think of it less as targeting a single pathway, and more as upgrading the infrastructure that keeps the whole system stable under load.
A Few Levers
Beyond training, several factors influence how this system behaves.
Adequate vitamin B6 intake supports key enzymatic steps within the kynurenine pathway, while sleep and stress management matter because glucocorticoids and inflammatory signals directly regulate IDO and TDO. Magnesium is important for general metabolic function, but the specific claim that it directly drives quinolinic acid-to-NAD+ conversion should be stated cautiously unless you want to cite a dedicated source.
None of these interventions is exotic. But they tend to act in the same direction — lowering background noise so the system does not default to stress-driven settings.
The Takeaway
The kynurenine pathway is not something to avoid — it is a fundamental component of human metabolism.
What matters is how it is regulated.
Under chronic stress and inflammation, this pathway shifts toward metabolites that can alter brain function in ways associated with depressive symptoms.
Training, nutrition, and recovery do not eliminate this pathway, but they can influence its direction.
In that sense, improving mental resilience is not only a matter of psychology.
It is, at least in part, a matter of metabolism.
And that metabolism is shaped — quite literally — by how you train.
References
- Agudelo et al., 2014. Skeletal muscle PGC-1α1 modulates kynurenine metabolism and mediates resilience to depression. Cell, 159(2), 33–45. [PMID: 25259918]pubmed.ncbi.nlm.nih
- Badawy, 2017. Kynurenine pathway of tryptophan metabolism: regulatory and functional aspects. Int. J. Tryptophan Res., 10, 1178646917691933. [PMID: 28469468]pubmed.ncbi.nlm.nih
- Cipriani et al., 2018. Comparative efficacy and acceptability of 21 antidepressant drugs for the acute treatment of adults with major depressive disorder: a systematic review and network meta-analysis. Lancet, 391(10128), 1357–1366. [PMID: 29477251]thelancet
- Dantzer et al., 2008. From inflammation to sickness and depression: when the immune system subjugates the brain. Nat. Rev. Neurosci., 9(1), 46–56. [PMID: 18094780]pmc.ncbi.nlm.nih
- Guillemin, 2012. Quinolinic acid, the inescapable neurotoxin. FEBS J., 279(8), 1356–1360. [PMID: 22339733]journals.sagepub
- O'Connor et al., 2009. Lipopolysaccharide-induced depressive-like behavior is mediated by the kynurenine pathway. Brain Behav. Immun., 23(4), 537–545. [PMID: 19486879]papersflow
- Raison et al., 2006. Cytokines sing the blues: inflammation and the pathogenesis of depression. Trends Immunol., 27(1), 24–31. [PMID: 16316783]academic.oup
- Rush et al., 2006. Acute and longer-term outcomes in depressed outpatients requiring one or several treatment steps: a STARD report. Am. J. Psychiatry*, 163(11), 1905–1917. [PMID: 17074942]pubmed.ncbi.nlm.nih
- Schwarcz et al., 2012. Kynurenines in the mammalian brain: when physiology meets pathology. Nat. Rev. Neurosci., 13(7), 465–477. [PMID: 22678511]nature
- Stone & Darlington, 2002. Endogenous kynurenines as targets for drug discovery and development. Nat. Rev. Drug Discov., 1(8), 609–620. [PMID: 12402501]pubmed.ncbi.nlm.nih