Sarcopenia and Strength Training: How to Stop Muscle Loss
Sarcopenia: When Muscle Metabolism Digs Its Heels Into Catabolism
Muscle does not disappear overnight, despite what you might think — yes, I’m looking at you, bodybuilders. More often than not, muscle fades slowly, almost shyly, and this, over decades.
For most of adulthood, skeletal muscle exists in a nice equilibrium. Proteins are broken down and rebuilt at roughly the same rate. Motor units fire reliably. Daily movement and training provide just enough mechanical stress to remind the body that muscle tissue—costly as it is—is still worth maintaining.
With age, however, that balance begins to drift ever so slightly. Just enough that the system starts operating with a catabolic bias.
Protein breakdown edges slightly ahead of synthesis. Recovery signals become weaker. And, neural input loses precision. Eventually, as years go by, the tissue remodels itself toward something smaller, slower, and less powerful.
This long, quiet drift is what we call sarcopenia. And biologically speaking, it is surprisingly complex. It is also tightly regulated.
To understand why muscle shrinks, we need to look at the systems that gradually push it in that direction.
Why Aging Muscle Shrinks
Sarcopenia is not caused by a defect or a single isolated event. It is the convergence of several physiological changes that all affect muscle tissue.
Among the most consistently identified contributors are:
- loss of motor neurons
- preferential atrophy of fast-twitch fibers
- mitochondrial dysfunction
- chronic low-grade inflammation
- hormonal shifts
- reduced sensitivity to anabolic signals
Taken individually, each factor might only nudge the system, but together they gradually tilt the body toward muscle breakdown and away from renewal.
Modern reviews emphasize that aging muscle stems from the interaction of neural, metabolic, and inflammatory processes, rather than a single faulty or failing pathway (Damanti et al., 2025; Liu et al., 2024).
Put differently: the machinery of muscle growth does not disappear with age.
It simply becomes sluggish and a bit more stubborn.
One of the earliest and most noticeable consequences of this shift is a decline in muscle power.
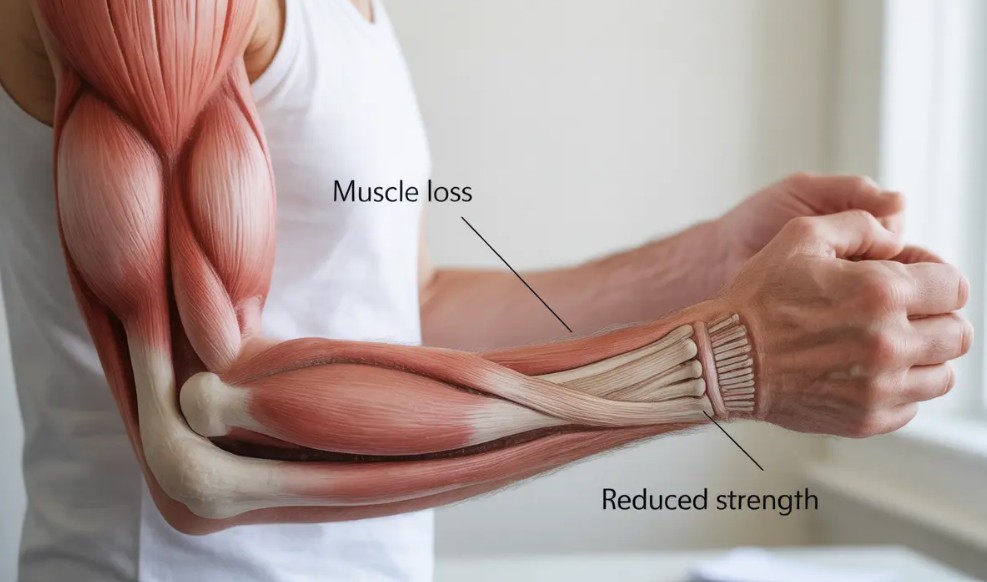
Muscle Power Is the First Thing to Go
Muscle power declines faster than maximal strength, and this is one of the most consistent observations in aging physiology literature.
After midlife, power may drop roughly two to three times faster than strength itself (Macaluso & De Vito, 2004).
The explanation resides largely in muscle fiber biology. You know, that human skeletal muscle contains a mixture of slow-twitch (type I) and fast-twitch (type II) fibers. The latter are responsible for rapid force production—jumping, sprinting, and also catching yourself during a stumble or a fall. Unfortunately, these are the very fibers that are the most vulnerable to aging.
In sedentary older adults, the cross‑sectional area of type II muscle fibers is reduced by roughly 25-35%, while type I fibers are relatively preserved (Dowling et al., 2023).
The result is a muscle that can still produce force… but does so more slowly. From a functional perspective, this matters A LOT. Many everyday movements such as climbing stairs, regaining balance, or preventing a fall depend on how quickly force can be produced, not just on maximal strength.
Reassuringly and interestingly, older adults who have engaged in lifelong resistance training often show a far better ability to preserve type II fiber morphology (Tøien et al., 2023).
So, the take home message is that this decline is not inevitable. It is, at the very least - negotiable.
Part of that decline, however, does not originate in the muscle itself.
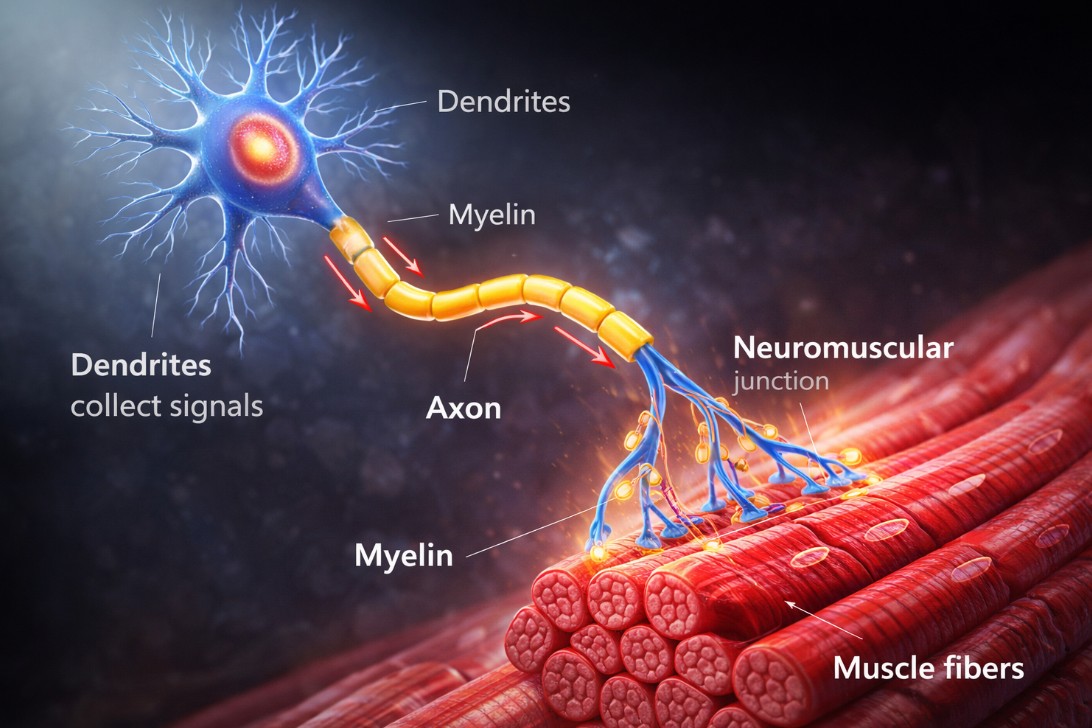
The Nervous System Is Aging Too
Muscle tissue rarely gets all the attention in these discussions, and yet it deserves at least some of it.
Motor neurons gradually die with age (up to ~40% loss by the age of 70). As they disappear, the number of motor units available to activate muscle fibers declines.
Some surviving neurons attempt to compensate by reinnervating abandoned fibers, creating larger motor units. For a while, this can work reasonably well.
But this compensation comes at a cost: fewer motor units overall, larger and less stable remaining units, and an increasing pool of denervated fibers.
The result is a system that produces less maximal force, with poorer coordination—and, most importantly, a reduced ability to generate force quickly (power) (Hunter, 2025).
From a training perspective, this neural component explains why high-velocity resistance training—lifting with intentional speed—can be particularly valuable in older adults.
Muscle strength matters, of course. But so does keeping the wiring intact. Use it—or lose it… as the saying goes
And even when the muscle is properly activated, another limitation begins to emerge: the muscle becomes less responsive to growth signals.
Anabolic Resistance: The Signal Needs to Be Louder
Another hallmark of aging muscle is something researchers call anabolic resistance.
In younger adults, a relatively modest protein intake—around 20–25 grams (≈0.24 g/kg) in a meal—can stimulate near-maximal muscle protein synthesis.
Older muscle generally requires a stronger stimulus, often closer to 30–40 grams of high-quality protein (≈0.4 g/kg), to produce a comparable response (Breen & Phillips, 2011; Moore et al., 2014; Smeuninx et al., 2017).
Mechanistically, this reflects a reduced sensitivity of the system: key growth pathways such as Akt–mTOR signaling are less responsive, and ribosomal biogenesis following feeding or resistance exercise is blunted (Brook et al., 2016; Chaillou & Montiel-Rojas, 2023).
The machinery is still there. It just needs a stronger signal.
From a coaching perspective, the implication is straightforward: if the signal must be louder, then the program must oblige.
Slightly higher protein intakes—distributed across meals—and consistent resistance training can substantially improve, and often restore, a robust anabolic response in older adults.
Part of what drives this reduced sensitivity is the broader physiological environment in which the muscle operates—particularly chronic low-grade inflammation.
Inflammation and the Catabolic Environment
Aging is often accompanied by a state of chronic low-grade inflammation, sometimes referred to as inflammaging.
Circulating inflammatory cytokines act as biochemical “stress signals.” Inside muscle cells, they activate pathways such as NF-κB—which promotes protein breakdown—and create conditions that engage FOXO signaling, further accelerating the recycling of muscle proteins through the ubiquitin–proteasome system.
At the same time, these signals interfere with anabolic pathways. In practical terms, this means the muscle becomes less responsive to growth signals like protein intake or resistance training, partly through impaired IGF-1/Akt signaling (Draganidis et al., 2021; Haberecht-Müller et al., 2021).
The result is not dramatic muscle wasting, but a persistent shift in daily protein turnover toward the catabolic side.
Lifestyle factors that reduce systemic inflammation—regular exercise, adequate sleep, and good metabolic health—therefore support muscle maintenance in ways that are not always immediately obvious.
Physiology is rarely compartmentalized.
Given this complexity, diagnosing sarcopenia cannot rely on a single variable.
Diagnosing Sarcopenia: Function First
In clinical practice, sarcopenia is no longer defined primarily by muscle mass.
Modern diagnostic frameworks place muscle strength at the center. Low muscle quantity or quality confirms the diagnosis. And impaired physical performance is used to grade its severity.
In practical terms, the sequence is usually simple: first assess muscle strength, then confirm low muscle mass, and finally evaluate physical performance.
Strength comes first because it predicts disability, loss of independence, and mortality more reliably than muscle mass alone (Cruz-Jentoft et al., 2018; Chen et al., 2020).
This reflects a simple truth: muscle does not matter because it is there. It matters because it keeps people capable.
Which naturally leads to the next question: how do we preserve that capacity?
Resistance Training: Still the Best Medicine
Despite the intricate biology underlying sarcopenia, the most reliable intervention remains strikingly uncomplicated: resistance training.
Large trials and meta-analyses consistently show that older adults—even those well into their eighties and beyond—can gain meaningful strength, muscle mass, and physical function when exposed to progressive resistance exercise (Grgic et al., 2020; Marzuca-Nassr et al., 2023).
Current position statements recommend programs built around a few durable principles:
- training two to three times per week
- targeting the major muscle groups
- applying progressive overload
- including movements performed with intent and, when appropriate, speed
These programs do more than increase muscle size. They also improve balance, mobility, functional capacity, and independence (Fragala et al., 2019; Rodrigues et al., 2022).
For all the molecular complexity upstream, the practical message downstream is simple: muscle still responds when it is challenged.
Once the fundamentals are in place, the natural question becomes: do supplements add anything meaningful?
A Brief Word on HMB
Few supplements in the muscle-loss conversation have generated as much debate as β-hydroxy-β-methylbutyrate (HMB).
HMB is a metabolite of the amino acid leucine and has been proposed to both reduce muscle protein breakdown and stimulate anabolic signaling.
Mechanistically, the rationale is sound: HMB can influence mTOR activity and attenuate proteolytic pathways, potentially shifting muscle protein turnover toward a more anabolic balance.
Clinical outcomes, however, are less impressive than supplement marketing might suggest.
Some studies report modest improvements in muscle mass or strength in frail older adults, hospitalized patients, or individuals undergoing prolonged inactivity.
In healthy, well-nourished individuals—especially those already resistance training—the additional benefit appears small or negligible.
Most reviews converge on the same conclusion: HMB may have a role in specific, catabolic contexts, but its effects are modest and do not rival those of resistance training and adequate protein intake (Kang, 2025).
Which is a long way of saying: supplements don’t replace fundamentals—they only amplify them when those are already in place.
So what do those fundamentals look like in practice?
A Practical Checklist for Aging Lifters
For those interested in preserving muscle as the decades accumulate, the research points toward a handful of dependable principles.
Lift regularly. Two to four resistance sessions per week targeting the major muscle groups is a solid baseline.
Eat enough protein. Roughly 0.4 g/kg per meal, distributed across the day, is a useful target for older adults.
Move explosively when appropriate. Fast concentric phases may help preserve power and motor-unit recruitment, especially when performed safely and with intent.
Stay generally active. Sedentary behavior accelerates muscle loss, while habitual movement helps preserve strength and function.
Look after recovery. Sleep, metabolic health, and body composition all help shape the anabolic environment.
None of these practices are revolutionary. They are simply consistent. And their impact over time is anything but trivial.
Aging Muscle Is Still Adaptable
Sarcopenia is often described as an unavoidable part of aging.
That description is technically correct, but also slightly misleading.
The rate and severity of muscle loss vary enormously between individuals. Older adults who remain physically active, consume adequate protein, and maintain good metabolic health often retain far more muscle mass and functional capacity than their sedentary peers (Paulussen et al., 2021; Horwath et al., 2024).
In other words, aging muscle may require stronger signals—but it remains remarkably responsive.
Sarcopenia is not a sudden collapse. It is the slow arithmetic of too little stimulus, repeated for too many years.
Fortunately, the body is still willing to respond when the inputs improve.